Request a demo specialized to your need.
The pharmacovigilance landscape is undergoing a fundamental shift. Regulatory scrutiny is intensifying, adverse event volumes are climbing, and the window between signal detection and regulatory submission is narrowing. Legacy safety systems — siloed, on-premise, and built for a different era — are buckling under the pressure.
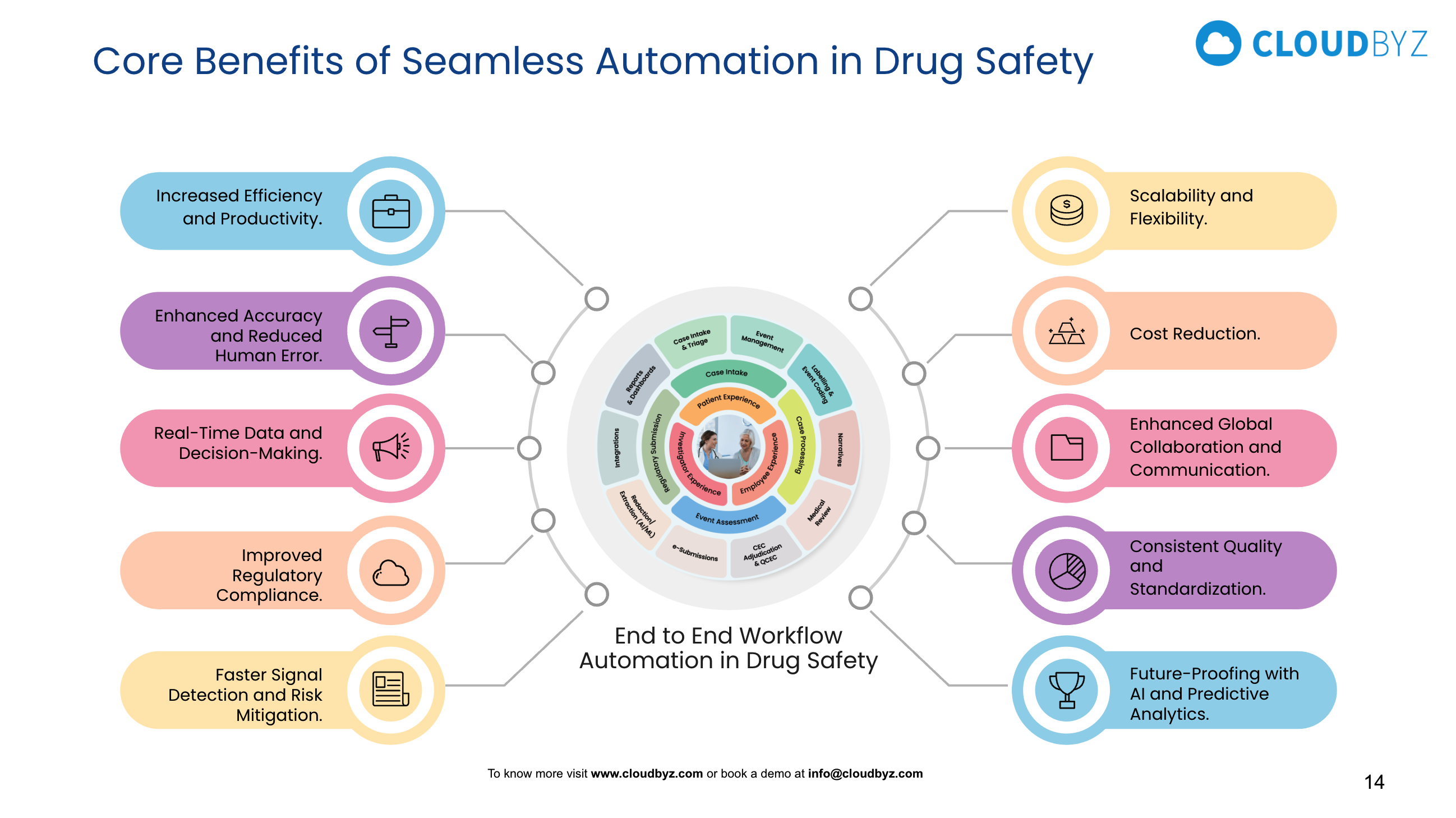
Cloudbyz Safety & Pharmacovigilance is a Salesforce-native platform purpose-built to modernize how pharma companies, biotechs, and CROs manage drug safety across the full product lifecycle. Here is a detailed look at what makes it stand out.
1. Built Natively on Salesforce — Not Just Integrated
Most safety platforms bolt on a Salesforce connector as an afterthought. Cloudbyz Safety is architected from the ground up on the Salesforce platform, which means it inherits the full Salesforce stack: security model, audit trails, workflow engine, reporting layer, and AppExchange ecosystem. There is no middleware to maintain, no API fragility to manage, and no version-lock headaches. IT teams already familiar with Salesforce administration can govern the safety platform without specialized vendor support.
2. End-to-End Adverse Event Management
From initial intake through case closure, Cloudbyz Safety manages the entire adverse event lifecycle in a single system. Individual Case Safety Reports (ICSRs) can be created from multiple intake channels — web forms, email, call center transcripts, and partner submissions — and are automatically routed to the appropriate workflow queue. Case processing steps including narrative writing, medical coding, causality assessment, and regulatory determination are tracked, timestamped, and auditable at every stage.
3. MedDRA and WHODrug Coding Integration
Accurate coding is non-negotiable in pharmacovigilance. Cloudbyz Safety integrates with MedDRA and WHODrug dictionaries, enabling coders to search, suggest, and apply standardized terminology directly within the case record. Auto-suggest functionality reduces manual lookup time and coding inconsistency, while coding audit trails ensure every change is traceable — a requirement under ICH E2B and FDA 21 CFR Part 11.
4. Regulatory Submission Ready: E2B(R3), EudraVigilance, FDA FAERS
Cloudbyz Safety is designed to meet current global submission standards. The platform supports E2B(R3) XML generation for EudraVigilance and FDA FAERS submissions. Expedited reports — 7-day and 15-day ICSRs for serious unexpected adverse reactions — are tracked against submission deadlines with automated escalation alerts to prevent regulatory breaches. For organizations operating across multiple geographies, the platform manages jurisdiction-specific submission timelines from a unified case record.
5. Signal Detection and Management
Pharmacovigilance is not just about reporting — it is about identifying safety signals before they become regulatory crises. Cloudbyz Safety includes signal detection workflows that aggregate case data, flag disproportionality, and surface potential signals for medical review. Safety teams can document signal assessments, assign follow-up actions, and track signal disposition through closure, creating a defensible record of proactive safety surveillance.
6. Periodic Safety Report Workflows: PSUR, PBRER, DSUR
Aggregate safety reporting is among the most resource-intensive obligations in pharmacovigilance. Cloudbyz Safety provides structured workflows for Periodic Safety Update Reports (PSURs), Periodic Benefit-Risk Evaluation Reports (PBRERs), and Development Safety Update Reports (DSURs). Templates, data pull configurations, and review-approval workflows are built into the platform, reducing the cycle time required to compile, review, and submit these reports against ICH and regional deadlines.
7. Risk Management Plan (RMP) Tracking
Regulatory authorities increasingly require robust Risk Management Plans as a condition of product approval. Cloudbyz Safety connects safety case data to RMP obligations, enabling teams to monitor the effectiveness of risk minimization measures in near real-time. Planned versus actual milestones for RMP commitments are tracked within the same platform, giving pharmacovigilance and regulatory affairs teams shared visibility into compliance status.
8. Literature Monitoring and Case Intake from Published Sources
Published medical literature is a mandated source of safety information under EMA and FDA guidelines, yet it is often managed through disconnected spreadsheets and email threads. Cloudbyz Safety supports structured literature monitoring workflows, enabling safety teams to screen publications, document relevance assessments, and convert qualifying literature cases into ICSRs without leaving the platform. This closes one of the most common audit gaps in pharmacovigilance operations.
9. Partner and Investigator Safety Reporting Portal
For companies running clinical trials or managing commercial partners, Cloudbyz Safety provides a configurable external-facing reporting portal. Investigators, CROs, licensees, and co-promotion partners can submit safety reports directly through a secure web interface, which feeds directly into the case management workflow. Automated acknowledgment letters and follow-up request tracking reduce the back-and-forth typically managed over email, compressing case intake timelines.
10. Real-Time Dashboards and Compliance Metrics
Safety leadership needs immediate visibility into case volumes, aging, submission compliance, and signal status — not monthly PowerPoint reports. Cloudbyz Safety delivers configurable real-time dashboards powered by Salesforce's native analytics engine. KPIs such as overdue cases, submission compliance rates by product and market, and case processing cycle times are available at a glance. These dashboards are role-specific, so case processors, medical reviewers, and the QPPV each see the information most relevant to their function.
11. Full Audit Trail and 21 CFR Part 11 Compliance
Regulatory agencies expect a complete, unaltered record of every action taken on a safety case. Cloudbyz Safety's audit trail captures every field-level change, every user action, and every system event with immutable timestamps. Combined with Salesforce's native electronic signature framework, the platform satisfies FDA 21 CFR Part 11 and EU Annex 11 requirements for electronic records and signatures — removing a major validation burden for compliance and QA teams.
12. Unified with Cloudbyz CTMS and eTMF for Integrated Clinical Safety
Perhaps the most strategically significant advantage: Cloudbyz Safety is part of a unified Salesforce-native eClinical suite that includes CTMS, eTMF, EDC, and Clinical Trial Financial Management. Safety data does not live in a separate silo — it is connected to the trial record, the subject enrollment data, the investigator profile, and the trial master file. This integration enables cross-functional safety reviews, accelerates regulatory inspection readiness, and gives clinical operations and pharmacovigilance a shared source of truth for the first time.
The Bottom Line
Pharmacovigilance is a discipline where operational failure carries direct patient safety consequences and regulatory penalties. The platforms that support it should be held to the same standard: reliable, auditable, integrated, and built for scale. Cloudbyz Safety delivers on that standard by combining the configurability of a modern SaaS platform with the enterprise-grade infrastructure of Salesforce — and by connecting safety operations to the broader clinical and regulatory ecosystem rather than treating them as an afterthought.
For pharma companies, biotechs, and CROs ready to replace disconnected legacy systems with a platform that grows with their pipeline, Cloudbyz Safety represents a compelling path forward.
Cloudbyz is the only unified eClinical platform built natively on Salesforce, offering CTMS, eTMF, EDC, Safety & PV, and AI-powered capabilities in a single environment. Learn more at cloudbyz.com.
Subscribe to our Newsletter

